Валентность — это способность атомов химических элементов образовывать определенное число химических связей с атомами других химических элементов.
Ковалентные связи могут образовываться по обменному и донорно-акцепторному механизмам.
Обменный механизм образования ковалентной связи — в образовании связи участвуют одноэлектронные атомные орбитали, т.е. каждый из атомов предоставляет по одному неспаренному электрону.
![]()
Донорно-акцепторный механизм — образование связи происходит за счет электронной пары одного из атомов (атом-донор) и вакантной орбитали другого атома (атом-акцептор):
![]()
Таким образом, атомы могут образовывать химическую связь не только за счет неспаренных электронов на внешнем энергетическом уровне, но и за счет неподеленных электронных пар, или свободных орбиталей на этом уровне.
Большинство элементов характеризуются высшей, низшей или промежуточной валентностью в соединениях.
Для большинства элементов высшая валентность, как правило, равна номеру группы, низшая валентность определяется по формуле: 8 — № группы. Промежуточная валентность – это число между низшей и высшей валентностями.
Например, высшая валентность хлора равна VII, низшая валентность хлора равна I, промежуточные валентности — III, V.
Обратите внимание! Степень окисления и валентность — это не одно и то же. Хотя иногда степени окисления совпадают с валентностями. Стпень окисления — это условный заряд атома, он может быть и положительным и отрицательным. А вот образовать отрицательное число связей атом никак не может.
Например, валентность (число связей) атома кислорода в молекуле O2 равна II, а вот степень окисления атома кислорода равна 0.

Большинство элементов проявляют переменную валентность в соединениях, но некоторые элементы проявляют постоянную валентность. Их необходимо запомнить:
| Элемент | Валентность |
| Фтор F | I |
| Кислород О | II |
| Металлы IA группы (Li, Na, K, Rb, Cs, Fr) | I |
| Металлы IIA группы (Be, Mg, Ca, Sr, Ba, Ra) | II |
| Алюминий Al | III |

Как определить валентность атома в соединении?
Рассмотрим валентные возможности атомов второго периода. В силу некоторых ограничений они не соответствуют традиционным «школьным» представлениям.
Итак, не внешнем энергетическом уровне лития 1 неспаренный электрон: 1s22s1.
+3Li 1s2 ![]() 2s1
2s1 ![]()
Следовательно, литий может образовывать одну связь и валентность лития I.
У бериллия на внешнем энергетическом уровне 2 электрона: 1s22s2.
+4Be 1s2 ![]() 2s2
2s2 ![]()
В возбужденном состоянии возможен переход электронов внешнего энергетического уровня с одного подуровня на другой: 1s22s12p1.
+4Be* 1s2 ![]() 2s1
2s1 ![]() 2p1
2p1![]()
Таким образом, на внешнем энергетическом уровне бериллия в возбужденном энергетическом состоянии есть 2 неспаренных электрона и две вакантные электронные орбитали. Следовательно, бериллий может образовать 2 связи по обменному механизму, т.е. валентность бериллия равна номеру группы и равна II.
Например, в хлориде бериллия валентность бериллия равна II:

Электронная конфигурация атома бора в основном состоянии +5B 1s22s22p1:
+5B 1s2 ![]() 2s2
2s2 ![]() 2p1
2p1![]()
В возбужденном состоянии: +5B* 1s22s12p2.
+5B 1s2 ![]() 2s1
2s1 ![]() 2p2
2p2![]()
Следовательно, бор может образовывать 3 связи по обменному механизму (за счет неспаренных электронов). Валентность бора в соединениях — III.
Например, в трихлориде бора BCl3 валентность бора равна III.

Однако, при этом у бора остается еще одна вакантная электронная орбиталь. Следовательно, бор может выступать, как акцептор электронной пары.
У атома углерода в возбужденном состоянии на внешнем энергетическом уровне 4 неспаренных электрона: 1s22s12p3, следовательно, максимальная валентность углерода равна IV (как правило, в органических соединениях у углерода именно такая валентность). В основном состоянии у атома углерода 2 неспаренных электрона, и валентность II. Однако посмотрим внимательно: у атома углерода в основном состоянии не внешнем энергетическом уровне есть незанятая (вакантная) электронная орбиталь. Следовательно, он может образовывать еще одну связь — по донорно-акцепторному механизму. Таким образом, в некоторых случаях углерод может образовывать три связи (например, молекула угарного газа CO, строение которой мы рассмотрим позднее).
Валентные возможности атома азота определяются также строением его внешнего энергетического уровня. В основном состоянии электронная формула азота: +7N 1s22s22p3.
За счет 3 неспаренных электронов на p-подуровне азот может образовывать 3 связи по обменному механизму (валентность III), и еще 1 связь азот может образовать по донорно-акцепторному механизму за счет неподеленной электронной пары. Таким образом, максимальная валентность азота в соединениях — IV. На примере азота можно убедиться, что высшая валентность атома и максимальная степень окисления — разные величины, которые далеко не всегда совпадают. Возбужденное состояние с 5 неспаренными электронами для атома азота не реализуется, т.к. на 2 энергетическом уровне есть только s и p орбитали.
Валентность химических элементов (Таблица)
Валентность химических элементов – это способность у атомов хим. элементов образовывать некоторое число химических связей. Принимает значения от 1 до 8 и не может быть равна 0. Определяется числом электронов атома затраченых на образование хим. связей с другим атомом. Валентность это реальная величина. Обозначается римскими цифрами (I ,II, III, IV, V, VI, VII, VIII).
Как можно определить валентность в соединениях:
— Валентность водорода (H) постоянна всегда 1. Отсюда в соединении H2O валентность O равна 2.
— Валентность кислорода (O) постоянна всегда 2. Отсюда в соединении СО2 валентность С равно 4.
— Высшая валентность всегда равна № группы.
— Низшая валентность равна разности между числом 8 (количество групп в Таблице Менделеева) и номером группы, в которой находится элемент.
— У металлов в подгруппах А таблицы Менделеева, валентность = № группы.
— У неметаллов обычно две валентности: высшая и низшая.

Валентность химических элементов может быть постоянной и переменной. Постоянная в основном у металлов главных подгрупп, переменная у неметаллов и металлов побочных подгруп.
Таблица валентности химических элементов
| Атомный № | Химический элемент | Символ | Валентность химических элементов | Примеры соединений |
| 1 | Водород / Hydrogen | H | I | HF |
| 2 | Гелий / Helium | He | отсутствует | — |
| 3 | Литий / Lithium | Li | I | Li2O |
| 4 | Бериллий / Beryllium | Be | II | BeH2 |
| 5 | Бор / Boron | B | III | BCl3 |
| 6 | Углерод / Carbon | C | IV, II | CO2, CH4 |
| 7 | Азот / Nitrogen | N | I, II, III, IV | NH3 |
| 8 | Кислород / Oxygen | O | II | H2O, BaO |
| 9 | Фтор / Fluorine | F | I | HF |
| 10 | Неон / Neon | Ne | отсутствует | — |
| 11 | Натрий / Sodium | Na | I | Na2O |
| 12 | Магний / Magnesium | Mg | II | MgCl2 |
| 13 | Алюминий / Aluminum | Al | III | Al2O3 |
| 14 | Кремний / Silicon | Si | IV | SiO2, SiCl4 |
| 15 | Фосфор / Phosphorus | P | III, V | PH3, P2O5 |
| 16 | Сера / Sulfur | S | VI, IV, II | H2S, SO3 |
| 17 | Хлор / Chlorine | Cl | I, III, V, VII | HCl, ClF3 |
| 18 | Аргон / Argon | Ar | отсутствует | — |
| 19 | Калий / Potassium | K | I | KBr |
| 20 | Кальций / Calcium | Ca | II | CaH2 |
| 21 | Скандий / Scandium | Sc | III | Sc2S3 |
| 22 | Титан / Titanium | Ti | II, III, IV | Ti2O3, TiH4 |
| 23 | Ванадий / Vanadium | V | II, III, IV, V | VF5, V2O3 |
| 24 | Хром / Chromium | Cr | II, III, VI | CrCl2, CrO3 |
| 25 | Марганец / Manganese | Mn | II, III, IV, VI, VII | Mn2O7, Mn2(SO4)3 |
| 26 | Железо / Iron | Fe | II, III | FeSO4, FeBr3 |
| 27 | Кобальт / Cobalt | Co | II, III | CoI2, Co2S3 |
| 28 | Никель / Nickel | Ni | II, III, IV | NiS, Ni(CO)4 |
| 29 | Медь / Copper | Сu | I, II | CuS, Cu2O |
| 30 | Цинк / Zinc | Zn | II | ZnCl2 |
| 31 | Галлий / Gallium | Ga | III | Ga(OH)3 |
| 32 | Германий / Germanium | Ge | II, IV | GeBr4, Ge(OH)2 |
| 33 | Мышьяк / Arsenic | As | III, V | As2S5, H3AsO4 |
| 34 | Селен / Selenium | Se | II, IV, VI, | H2SeO3 |
| 35 | Бром / Bromine | Br | I, III, V, VII | HBrO3 |
| 36 | Криптон / Krypton | Kr | VI, IV, II | KrF2, BaKrO4 |
| 37 | Рубидий / Rubidium | Rb | I | RbH |
| 38 | Стронций / Strontium | Sr | II | SrSO4 |
| 39 | Иттрий / Yttrium | Y | III | Y2O3 |
| 40 | Цирконий / Zirconium | Zr | II, III, IV | ZrI4, ZrCl2 |
| 41 | Ниобий / Niobium | Nb | I, II, III, IV, V | NbBr5 |
| 42 | Молибден / Molybdenum | Mo | II, III, IV, V, VI | Mo2O5, MoF6 |
| 43 | Технеций / Technetium | Tc | I — VII | Tc2S7 |
| 44 | Рутений / Ruthenium | Ru | II — VIII | RuO4, RuF5, RuBr3 |
| 45 | Родий / Rhodium | Rh | I, II, III, IV, V | RhS, RhF3 |
| 46 | Палладий / Palladium | Pd | I, II, III, IV | Pd2S, PdS2 |
| 47 | Серебро / Silver | Ag | I, II, III | AgO, AgF2, AgNO3 |
| 48 | Кадмий / Cadmium | Cd | II | CdCl2 |
| 49 | Индий / Indium | In | III | In2O3 |
| 50 | Олово / Tin | Sn | II, IV | SnBr4, SnF2 |
| 51 | Сурьма / Antimony | Sb | III, V | SbF5, SbH3 |
| 52 | Теллур / Tellurium | Te | VI, IV, II | TeH2, H6TeO6 |
| 53 | Иод / Iodine | I | I, III, V, VII | HIO3, HI |
| 54 | Ксенон / Xenon | Xe | II, IV, VI, VIII | XeF6, XeO4, XeF2 |
| 55 | Цезий / Cesium | Cs | I | CsCl |
| 56 | Барий / Barium | Ba | II | Ba(OH)2 |
| 57 | Лантан / Lanthanum | La | III | LaH3 |
| 58 | Церий / Cerium | Ce | III, IV | CeO2 , CeF3 |
| 59 | Празеодим / Praseodymium | Pr | III, IV | PrF4, PrO2 |
| 60 | Неодим / Neodymium | Nd | III | Nd2O3 |
| 61 | Прометий / Promethium | Pm | III | Pm2O3 |
| 62 | Самарий / Samarium | Sm | II, III | SmO |
| 63 | Европий / Europium | Eu | II, III | EuSO4 |
| 64 | Гадолиний / Gadolinium | Gd | III | GdCl3 |
| 65 | Тербий / Terbium | Tb | III, IV | TbF4, TbCl3 |
| 66 | Диспрозий / Dysprosium | Dy | III | Dy2O3 |
| 67 | Гольмий / Holmium | Ho | III | Ho2O3 |
| 68 | Эрбий / Erbium | Er | III | Er2O3 |
| 69 | Тулий / Thulium | Tm | II, III | Tm2O3 |
| 70 | Иттербий / Ytterbium | Yb | II, III | YO |
| 71 | Лютеций / Lutetium | Lu | III | LuF3 |
| 72 | Гафний / Hafnium | Hf | II, III, IV | HfBr3, HfCl4 |
| 73 | Тантал / Tantalum | Ta | I — V | TaCl5, TaBr2, TaCl4 |
| 74 | Вольфрам / Tungsten | W | II — VI | WBr6, Na2WO4 |
| 75 | Рений / Rhenium | Re | I — VII | Re2S7, Re2O5 |
| 76 | Осмий / Osmium | Os | II — VI, VIII | OsF8, OsI2, Os2O3 |
| 77 | Иридий / Iridium | Ir | I — VI | IrS3, IrF4 |
| 78 | Платина / Platinum | Pt | I, II, III, IV, V | Pt(SO4)3, PtBr4 |
| 79 | Золото / Gold | Au | I, II, III | AuH, Au2O3, Au2Cl6 |
| 80 | Ртуть / Mercury | Hg | II | HgF2, HgBr2 |
| 81 | Талий / Thallium | Tl | I, III | TlCl3, TlF |
| 82 | Свинец / Lead | Pb | II, IV | PbS, PbH4 |
| 83 | Висмут / Bismuth | Bi | III, V | BiF5, Bi2S3 |
| 84 | Полоний / Polonium | Po | VI, IV, II | PoCl4, PoO3 |
| 85 | Астат / Astatine | At | нет данных | — |
| 86 | Радон / Radon | Rn | отсутствует | — |
| 87 | Франций / Francium | Fr | I | — |
| 88 | Радий / Radium | Ra | II | RaBr2 |
| 89 | Актиний / Actinium | Ac | III | AcCl3 |
| 90 | Торий / Thorium | Th | II, III, IV | ThO2, ThF4 |
| 91 | Проактиний / Protactinium | Pa | IV, V | PaCl5, PaF4 |
| 92 | Уран / Uranium | U | III, IV | UF4, UO3 |
| 93 | Нептуний | Np | III — VI | NpF6, NpCl4 |
| 94 | Плутоний | Pu | II, III, IV | PuO2, PuF3, PuF4 |
| 95 | Америций | Am | III — VI | AmF3, AmO2 |
| 96 | Кюрий | Cm | III, IV | CmO2, Cm2O3 |
| 97 | Берклий | Bk | III, IV | BkF3, BkO2 |
| 98 | Калифорний | Cf | II, III, IV | Cf2O3 |
| 99 | Эйнштейний | Es | II, III | EsF3 |
| 100 | Фермий | Fm | II, III | — |
| 101 | Менделевий | Md | II, III | — |
| 102 | Нобелий | No | II, III | — |
| 103 | Лоуренсий | Lr | III | — |
| Номер | Элемент | Символ | Валентность химических элементов | Пример |
Поделитесь ссылкой с друзьями:
Похожие таблицы
Комментарии:
In chemistry, the valence (US spelling) or valency (British spelling) of an element is the measure of its combining capacity with other atoms when it forms chemical compounds or molecules.
Description
The combining capacity, or affinity of an atom of a given element is determined by the number of hydrogen atoms that it combines with. In methane, carbon has a valence of 4; in ammonia, nitrogen has a valence of 3; in water, oxygen has a valence of 2; and in hydrogen chloride, chlorine has a valence of 1. Chlorine, as it has a valence of one, can be substituted for hydrogen. Phosphorus has a valence of 5 in phosphorus pentachloride, PCl5. Valence diagrams of a compound represent the connectivity of the elements, with lines drawn between two elements, sometimes called bonds, representing a saturated valency for each element.[1] The two tables below show some examples of different compounds, their valence diagrams, and the valences for each element of the compound.
| Compound | H2 Hydrogen | CH4 Methane | C3H8 Propane | C3H6 Propylene | C2H2 Acetylene |
|---|---|---|---|---|---|
| Diagram |  | 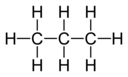 | 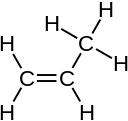 | ||
| Valencies |
|
|
|
|
|
| Compound | NH3 Ammonia | NaCN Sodium cyanide | PSCl3 Thiophosphoryl chloride | H2S Hydrogen sulfide | H2SO4 Sulfuric acid | H2S2O6 Dithionic acid | Cl2O7 Dichlorine heptoxide | XeO4 Xenon tetroxide |
|---|---|---|---|---|---|---|---|---|
| Diagram |  | 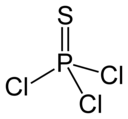 | 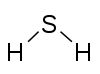 |  |  |  |  | |
| Valencies |
|
|
|
|
|
|
|
|
Modern definitions
Valence is defined by the IUPAC as:[2]
- The maximum number of univalent atoms (originally hydrogen or chlorine atoms) that may combine with an atom of the element under consideration, or with a fragment, or for which an atom of this element can be substituted.
An alternative modern description is:[3]
- The number of hydrogen atoms that can combine with an element in a binary hydride or twice the number of oxygen atoms combining with an element in its oxide or oxides.
This definition differs from the IUPAC definition as an element can be said to have more than one valence.
A very similar modern definition given in a recent article defines the valence of a particular atom in a molecule as «the number of electrons that an atom uses in bonding», with two equivalent formulas for calculating valence:[4]
- valence = number of electrons in valence shell of free atom – number of non-bonding electrons on atom in molecule,
and
- valence = number of bonds + formal charge.
Historical development
The etymology of the words valence (plural valences) and valency (plural valencies) traces back to 1425, meaning «extract, preparation», from Latin valentia «strength, capacity», from the earlier valor «worth, value», and the chemical meaning referring to the «combining power of an element» is recorded from 1884, from German Valenz.[5]

The concept of valence was developed in the second half of the 19th century and helped successfully explain the molecular structure of inorganic and organic compounds.[1]
The quest for the underlying causes of valence led to the modern theories of chemical bonding, including the cubical atom (1902), Lewis structures (1916), valence bond theory (1927), molecular orbitals (1928), valence shell electron pair repulsion theory (1958), and all of the advanced methods of quantum chemistry.
In 1789, William Higgins published views on what he called combinations of «ultimate» particles, which foreshadowed the concept of valency bonds.[6] If, for example, according to Higgins, the force between the ultimate particle of oxygen and the ultimate particle of nitrogen were 6, then the strength of the force would be divided accordingly, and likewise for the other combinations of ultimate particles (see illustration).
The exact inception, however, of the theory of chemical valencies can be traced to an 1852 paper by Edward Frankland, in which he combined the older radical theory with thoughts on chemical affinity to show that certain elements have the tendency to combine with other elements to form compounds containing 3, i.e., in the 3-atom groups (e.g., NO3, NH3, NI3, etc.) or 5, i.e., in the 5-atom groups (e.g., NO5, NH4O, PO5, etc.), equivalents of the attached elements. According to him, this is the manner in which their affinities are best satisfied, and by following these examples and postulates, he declares how obvious it is that[7]
A tendency or law prevails (here), and that, no matter what the characters of the uniting atoms may be, the combining power of the attracting element, if I may be allowed the term, is always satisfied by the same number of these atoms.
This “combining power” was afterwards called quantivalence or valency (and valence by American chemists).[6] In 1857 August Kekulé proposed fixed valences for many elements, such as 4 for carbon, and used them to propose structural formulas for many organic molecules, which are still accepted today.
Most 19th-century chemists defined the valence of an element as the number of its bonds without distinguishing different types of valence or of bond. However, in 1893 Alfred Werner described transition metal coordination complexes such as [Co(NH3)6]Cl3, in which he distinguished principal and subsidiary valences (German: ‘Hauptvalenz’ and ‘Nebenvalenz’), corresponding to the modern concepts of oxidation state and coordination number respectively.
For main-group elements, in 1904 Richard Abegg considered positive and negative valences (maximal and minimal oxidation states), and proposed Abegg’s rule to the effect that their difference is often 8.
Electrons and valence
The Rutherford model of the nuclear atom (1911) showed that the exterior of an atom is occupied by electrons, which suggests that electrons are responsible for the interaction of atoms and the formation of chemical bonds. In 1916, Gilbert N. Lewis explained valence and chemical bonding in terms of a tendency of (main-group) atoms to achieve a stable octet of 8 valence-shell electrons. According to Lewis, covalent bonding leads to octets by the sharing of electrons, and ionic bonding leads to octets by the transfer of electrons from one atom to the other. The term covalence is attributed to Irving Langmuir, who stated in 1919 that «the number of pairs of electrons which any given atom shares with the adjacent atoms is called the covalence of that atom».[8] The prefix co- means «together», so that a co-valent bond means that the atoms share a valence. Subsequent to that, it is now more common to speak of covalent bonds rather than valence, which has fallen out of use in higher-level work from the advances in the theory of chemical bonding, but it is still widely used in elementary studies, where it provides a heuristic introduction to the subject.
In the 1930s, Linus Pauling proposed that there are also polar covalent bonds, which are intermediate between covalent and ionic, and that the degree of ionic character depends on the difference of electronegativity of the two bonded atoms.
Pauling also considered hypervalent molecules, in which main-group elements have apparent valences greater than the maximal of 4 allowed by the octet rule. For example, in the sulfur hexafluoride molecule (SF6), Pauling considered that the sulfur forms 6 true two-electron bonds using sp3d2 hybrid atomic orbitals, which combine one s, three p and two d orbitals. However more recently, quantum-mechanical calculations on this and similar molecules have shown that the role of d orbitals in the bonding is minimal, and that the SF6 molecule should be described as having 6 polar covalent (partly ionic) bonds made from only four orbitals on sulfur (one s and three p) in accordance with the octet rule, together with six orbitals on the fluorines.[9] Similar calculations on transition-metal molecules show that the role of p orbitals is minor, so that one s and five d orbitals on the metal are sufficient to describe the bonding.[10]
Common valences
For elements in the main groups of the periodic table, the valence can vary between 1 and 7.
| Group | Valence 1 | Valence 2 | Valence 3 | Valence 4 | Valence 5 | Valence 6 | Valence 7 | Valence 8 | Typical valences |
|---|---|---|---|---|---|---|---|---|---|
| 1 (I) | NaCl | 1 | |||||||
| 2 (II) | MgCl2 | 2 | |||||||
| 13 (III) | BCl3 AlCl3 Al2O3 | 3 | |||||||
| 14 (IV) | CO | CH4 | 4 | ||||||
| 15 (V) | NO | NH3 PH3 As2O3 | NO2 | N2O5 PCl5 | 3 and 5 | ||||
| 16 (VI) | H2O H2S | SO2 | SO3 | 2 and 6 | |||||
| 17 (VII) | HCl | HClO2 | ClO2 | HClO3 | Cl2O7 | 1 and 7 | |||
| 18 (VIII) | XeO4 | 8 |
Many elements have a common valence related to their position in the periodic table, and nowadays this is rationalised by the octet rule.
The Greek/Latin numeral prefixes (mono-/uni-, di-/bi-, tri-/ter-, and so on) are used to describe ions in the charge states 1, 2, 3, and so on, respectively. Polyvalence or multivalence refers to species that are not restricted to a specific number of valence bonds. Species with a single charge are univalent (monovalent). For example, the Cs+ cation is a univalent or monovalent cation, whereas the Ca2+ cation is a divalent cation, and the Fe3+ cation is a trivalent cation. Unlike Cs and Ca, Fe can also exist in other charge states, notably 2+ and 4+, and is thus known as a multivalent (polyvalent) ion.[11] Transition metals and metals to the right are typically multivalent but there is no simple pattern predicting their valency.[12]
| Valence | More common adjective‡ | Less common synonymous adjective‡§ |
|---|---|---|
| 0-valent | zerovalent | nonvalent |
| 1-valent | monovalent | univalent |
| 2-valent | divalent | bivalent |
| 3-valent | trivalent | tervalent |
| 4-valent | tetravalent | quadrivalent |
| 5-valent | pentavalent | quinquevalent, quinquivalent |
| 6-valent | hexavalent | sexivalent |
| 7-valent | heptavalent | septivalent |
| 8-valent | octavalent | — |
| 9-valent | nonavalent | — |
| 10-valent | decavalent | — |
| 12-valent | dodecavalent | — |
| multiple / many / variable | polyvalent | multivalent |
| together | covalent | — |
| not together | noncovalent | — |
† The same adjectives are also used in medicine to refer to vaccine valence, with the slight difference that in the latter sense, quadri- is more common than tetra-.
‡ As demonstrated by hit counts in Google web search and Google Books search corpora (accessed 2017).
§ A few other forms can be found in large English-language corpora (for example, *quintavalent, *quintivalent, *decivalent), but they are not the conventionally established forms in English and thus are not entered in major dictionaries.
Valence versus oxidation state
Because of the ambiguity of the term valence,[13] other notations are currently preferred. Beside the system of oxidation states (also called oxidation numbers) as used in Stock nomenclature for coordination compounds,[14] and the lambda notation, as used in the IUPAC nomenclature of inorganic chemistry,[15] oxidation state is a more clear indication of the electronic state of atoms in a molecule.
The oxidation state of an atom in a molecule gives the number of valence electrons it has gained or lost.[16] In contrast to the valency number, the oxidation state can be positive (for an electropositive atom) or negative (for an electronegative atom).
Elements in a high oxidation state have an oxidation state higher than +4, and also, elements in a high valence state (hypervalent elements) have a valence higher than 4. For example, in perchlorates ClO−4, chlorine has 7 valence bonds (thus, it is heptavalent, in other words, it has valence 7), and it has oxidation state +7; in ruthenium tetroxide RuO4, ruthenium has 8 valence bonds (thus, it is octavalent, in other words, it has valence 8), and it has oxidation state +8.
In some scenarios, the difference between valence and oxidation state arises. Valence and oxidation state of the same atom may not be the same. For example, in disulfur decafluoride molecule S2F10, each sulfur atom has 6 valence bonds (5 single bonds with fluorine atoms and 1 single bond with sulfur atom), thus, each sulfur atom is hexavalent, in other words, it has valence 6, but has oxidation state +5. In dioxygen molecule O2, each oxygen atom has 2 valence bonds, thus, each oxygen atom is divalent, in other words, it has valence 2, but has oxidation state 0. In acetylene H−C≡C−H, each carbon atom has 4 valence bonds (1 single bond with hydrogen atom and 3 single bonds with carbon atom), thus, each carbon atom is tetravalent, in other words, it has valence 4, but has oxidation state −1.
Examples
| Compound | Formula | Valence | Oxidation state | Diagram |
|---|---|---|---|---|
| Hydrogen chloride | HCl | H = 1 Cl = 1 | H = +1 Cl = −1 | H−Cl |
| Perchloric acid * | HClO4 | H = 1 Cl = 7 O = 2 | H = +1 Cl = +7 O = −2 | 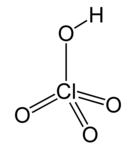 |
| Methane | CH4 | C = 4 H = 1 | C = −4 H = +1 | 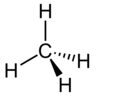 |
| Dichloromethane ** | CH2Cl2 | C = 4 H = 1 Cl = 1 | C = 0 H = +1 Cl = −1 | 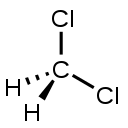 |
| Ferrous oxide *** | FeO | Fe = 2 O = 2 | Fe = +2 O = −2 | Fe=O |
| Ferric oxide *** | Fe2O3 | Fe = 3 O = 2 | Fe = +3 O = −2 | O=Fe−O−Fe=O |
| Sodium hydride | NaH | Na = 1 H = 1 | Na = +1 H = −1 | Na−H |
* The perchlorate ion ClO−4 is monovalent, in other words, it has valence 1.
** Valences may also be different from absolute values of oxidation states due to different polarity of bonds. For example, in dichloromethane, CH2Cl2, carbon has valence 4 but oxidation state 0.
*** Iron oxides appear in a crystal structure, so no typical molecule can be identified. In ferrous oxide, Fe has oxidation state +2; in ferric oxide, oxidation state +3.
| Compound | Formula | Valence | Oxidation state | Diagram |
|---|---|---|---|---|
| Hydrogen | H2 | H = 1 | H = 0 | H−H |
| Chlorine | Cl2 | Cl = 1 | Cl = 0 | Cl−Cl |
| Hydrogen peroxide | H2O2 | H = 1 O = 2 | H = +1 O = −1 |  |
| Hydrazine | N2H4 | H = 1 N = 3 | H = +1 N = −2 |  |
| Disulfur decafluoride | S2F10 | S = 6 F = 1 | S = +5 F = −1 |  |
| Dithionic acid | H2S2O6 | S = 6 O = 2 H = 1 | S = +5 O = −2 H = +1 |  |
| Hexachloroethane | C2Cl6 | C = 4 Cl = 1 | C = +3 Cl = −1 |  |
| Ethylene | C2H4 | C = 4 H = 1 | C = −2 H = +1 |  |
| Acetylene | C2H2 | C = 4 H = 1 | C = −1 H = +1 | H−C≡C−H |
| Mercury(I) chloride | Hg2Cl2 | Hg = 2 Cl = 1 | Hg = +1 Cl = −1 | Cl−Hg−Hg−Cl |
«Maximum number of bonds» definition
Frankland took the view that the valence (he used the term «atomicity») of an element was a single value that corresponded to the maximum value observed. The number of unused valencies on atoms of what are now called the p-block elements is generally even, and Frankland suggested that the unused valencies saturated one another. For example, nitrogen has a maximum valence of 5, in forming ammonia two valencies are left unattached; sulfur has a maximum valence of 6, in forming hydrogen sulphide four valencies are left unattached.[17][18]
The International Union of Pure and Applied Chemistry (IUPAC) has made several attempts to arrive at an unambiguous definition of valence. The current version, adopted in 1994:[19]
- The maximum number of univalent atoms (originally hydrogen or chlorine atoms) that may combine with an atom of the element under consideration, or with a fragment, or for which an atom of this element can be substituted.[2]
Hydrogen and chlorine were originally used as examples of univalent atoms, because of their nature to form only one single bond. Hydrogen has only one valence electron and can form only one bond with an atom that has an incomplete outer shell. Chlorine has seven valence electrons and can form only one bond with an atom that donates a valence electron to complete chlorine’s outer shell. However, chlorine can also have oxidation states from +1 to +7 and can form more than one bond by donating valence electrons.
Hydrogen has only one valence electron, but it can form bonds with more than one atom. In the bifluoride ion ([HF2]−), for example, it forms a three-center four-electron bond with two fluoride atoms:
- [F−H F− ↔ F− H−F]
Another example is the three-center two-electron bond in diborane (B2H6).
Maximum valences of the elements
Maximum valences for the elements are based on the data from list of oxidation states of the elements. They are shown by the color code at the bottom of the table.
Maximum valences of the elements | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | ||
| Group → | |||||||||||||||||||
| ↓ Period | |||||||||||||||||||
| 1 | 1 H | 2 He | |||||||||||||||||
| 2 | 3 Li | 4 Be | 5 B | 6 C | 7 N | 8 O | 9 F | 10 Ne | |||||||||||
| 3 | 11 Na | 12 Mg | 13 Al | 14 Si | 15 P | 16 S | 17 Cl | 18 Ar | |||||||||||
| 4 | 19 K | 20 Ca | 21 Sc | 22 Ti | 23 V | 24 Cr | 25 Mn | 26 Fe | 27 Co | 28 Ni | 29 Cu | 30 Zn | 31 Ga | 32 Ge | 33 As | 34 Se | 35 Br | 36 Kr | |
| 5 | 37 Rb | 38 Sr | 39 Y | 40 Zr | 41 Nb | 42 Mo | 43 Tc | 44 Ru | 45 Rh | 46 Pd | 47 Ag | 48 Cd | 49 In | 50 Sn | 51 Sb | 52 Te | 53 I | 54 Xe | |
| 6 | 55 Cs | 56 Ba | 71 Lu | 72 Hf | 73 Ta | 74 W | 75 Re | 76 Os | 77 Ir | 78 Pt | 79 Au | 80 Hg | 81 Tl | 82 Pb | 83 Bi | 84 Po | 85 At | 86 Rn | |
| 7 | 87 Fr | 88 Ra | 103 Lr | 104 Rf | 105 Db | 106 Sg | 107 Bh | 108 Hs | 109 Mt | 110 Ds | 111 Rg | 112 Cn | 113 Nh | 114 Fl | 115 Mc | 116 Lv | 117 Ts | 118 Og | |
| 57 La | 58 Ce | 59 Pr | 60 Nd | 61 Pm | 62 Sm | 63 Eu | 64 Gd | 65 Tb | 66 Dy | 67 Ho | 68 Er | 69 Tm | 70 Yb | ||||||
| 89 Ac | 90 Th | 91 Pa | 92 U | 93 Np | 94 Pu | 95 Am | 96 Cm | 97 Bk | 98 Cf | 99 Es | 100 Fm | 101 Md | 102 No | ||||||
| Maximum valences are based on the List of oxidation states of the elements | |||||||||||||||||||
|
Primordial From decay Synthetic Border shows natural occurrence of the element |
See also
- Abegg’s rule
- Oxidation state
References
- ^ a b Partington, James Riddick (1921). A text-book of inorganic chemistry for university students (1st ed.). OL 7221486M.
- ^ a b IUPAC Gold Book definition: valence
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ^ Parkin, Gerard (May 2006). «Valence, Oxidation Number, and Formal Charge: Three Related but Fundamentally Different Concepts». Journal of Chemical Education. 83 (5): 791. doi:10.1021/ed083p791. ISSN 0021-9584.
- ^ Harper, Douglas. «valence». Online Etymology Dictionary.
- ^ a b Partington, J.R. (1989). A Short History of Chemistry. Dover Publications, Inc. ISBN 0-486-65977-1.
- ^ Frankland, E. (1852). «On a New Series of Organic Bodies Containing Metals». Philosophical Transactions of the Royal Society of London. 142: 417–444. doi:10.1098/rstl.1852.0020. S2CID 186210604.
- ^ Langmuir, Irving (1919). «The Arrangement of Electrons in Atoms and Molecules». Journal of the American Chemical Society. 41 (6): 868–934. doi:10.1021/ja02227a002.
- ^ Magnusson, Eric (1990). «Hypercoordinate molecules of second-row elements: d functions or d orbitals?». J. Am. Chem. Soc. 112 (22): 7940–7951. doi:10.1021/ja00178a014.
- ^ Frenking, Gernot; Shaik, Sason, eds. (May 2014). «Chapter 7: Chemical bonding in Transition Metal Compounds». The Chemical Bond: Chemical Bonding Across the Periodic Table. Wiley – VCH. ISBN 978-3-527-33315-8.
- ^ Merriam-Webster, Merriam-Webster’s Unabridged Dictionary, Merriam-Webster, archived from the original on 2020-05-25, retrieved 2017-05-11.
- ^ «Lesson 7: Ions and Their Names». Clackamas Community College. Archived from the original on 21 January 2019. Retrieved 5 February 2019.
- ^ The Free Dictionary: valence
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the «Gold Book») (1997). Online corrected version: (2006–) «Oxidation number». doi:10.1351/goldbook.O04363
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the «Gold Book») (1997). Online corrected version: (2006–) «Lambda». doi:10.1351/goldbook.L03418
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the «Gold Book») (1997). Online corrected version: (2006–) «Oxidation state». doi:10.1351/goldbook.O04365
- ^ Frankland, E. (1870). Lecture notes for chemical students(Google eBook) (2d ed.). J. Van Voorst. p. 21.
- ^ Frankland, E.; Japp, F.R (1885). Inorganic chemistry (1st ed.). pp. 75–85. OL 6994182M.
- ^ Muller, P. (1994). «Glossary of terms used in physical organic chemistry (IUPAC Recommendations 1994)». Pure and Applied Chemistry. 66 (5): 1077–1184. doi:10.1351/pac199466051077. S2CID 195819485.
In chemistry, the valence (US spelling) or valency (British spelling) of an element is the measure of its combining capacity with other atoms when it forms chemical compounds or molecules.
Description
The combining capacity, or affinity of an atom of a given element is determined by the number of hydrogen atoms that it combines with. In methane, carbon has a valence of 4; in ammonia, nitrogen has a valence of 3; in water, oxygen has a valence of 2; and in hydrogen chloride, chlorine has a valence of 1. Chlorine, as it has a valence of one, can be substituted for hydrogen. Phosphorus has a valence of 5 in phosphorus pentachloride, PCl5. Valence diagrams of a compound represent the connectivity of the elements, with lines drawn between two elements, sometimes called bonds, representing a saturated valency for each element.[1] The two tables below show some examples of different compounds, their valence diagrams, and the valences for each element of the compound.
| Compound | H2 Hydrogen | CH4 Methane | C3H8 Propane | C3H6 Propylene | C2H2 Acetylene |
|---|---|---|---|---|---|
| Diagram |  |  |  | ||
| Valencies |
|
|
|
|
|
| Compound | NH3 Ammonia | NaCN Sodium cyanide | PSCl3 Thiophosphoryl chloride | H2S Hydrogen sulfide | H2SO4 Sulfuric acid | H2S2O6 Dithionic acid | Cl2O7 Dichlorine heptoxide | XeO4 Xenon tetroxide |
|---|---|---|---|---|---|---|---|---|
| Diagram |  |  |  |  |  |  |  | |
| Valencies |
|
|
|
|
|
|
|
|
Modern definitions
Valence is defined by the IUPAC as:[2]
- The maximum number of univalent atoms (originally hydrogen or chlorine atoms) that may combine with an atom of the element under consideration, or with a fragment, or for which an atom of this element can be substituted.
An alternative modern description is:[3]
- The number of hydrogen atoms that can combine with an element in a binary hydride or twice the number of oxygen atoms combining with an element in its oxide or oxides.
This definition differs from the IUPAC definition as an element can be said to have more than one valence.
A very similar modern definition given in a recent article defines the valence of a particular atom in a molecule as «the number of electrons that an atom uses in bonding», with two equivalent formulas for calculating valence:[4]
- valence = number of electrons in valence shell of free atom – number of non-bonding electrons on atom in molecule,
and
- valence = number of bonds + formal charge.
Historical development
The etymology of the words valence (plural valences) and valency (plural valencies) traces back to 1425, meaning «extract, preparation», from Latin valentia «strength, capacity», from the earlier valor «worth, value», and the chemical meaning referring to the «combining power of an element» is recorded from 1884, from German Valenz.[5]

The concept of valence was developed in the second half of the 19th century and helped successfully explain the molecular structure of inorganic and organic compounds.[1]
The quest for the underlying causes of valence led to the modern theories of chemical bonding, including the cubical atom (1902), Lewis structures (1916), valence bond theory (1927), molecular orbitals (1928), valence shell electron pair repulsion theory (1958), and all of the advanced methods of quantum chemistry.
In 1789, William Higgins published views on what he called combinations of «ultimate» particles, which foreshadowed the concept of valency bonds.[6] If, for example, according to Higgins, the force between the ultimate particle of oxygen and the ultimate particle of nitrogen were 6, then the strength of the force would be divided accordingly, and likewise for the other combinations of ultimate particles (see illustration).
The exact inception, however, of the theory of chemical valencies can be traced to an 1852 paper by Edward Frankland, in which he combined the older radical theory with thoughts on chemical affinity to show that certain elements have the tendency to combine with other elements to form compounds containing 3, i.e., in the 3-atom groups (e.g., NO3, NH3, NI3, etc.) or 5, i.e., in the 5-atom groups (e.g., NO5, NH4O, PO5, etc.), equivalents of the attached elements. According to him, this is the manner in which their affinities are best satisfied, and by following these examples and postulates, he declares how obvious it is that[7]
A tendency or law prevails (here), and that, no matter what the characters of the uniting atoms may be, the combining power of the attracting element, if I may be allowed the term, is always satisfied by the same number of these atoms.
This “combining power” was afterwards called quantivalence or valency (and valence by American chemists).[6] In 1857 August Kekulé proposed fixed valences for many elements, such as 4 for carbon, and used them to propose structural formulas for many organic molecules, which are still accepted today.
Most 19th-century chemists defined the valence of an element as the number of its bonds without distinguishing different types of valence or of bond. However, in 1893 Alfred Werner described transition metal coordination complexes such as [Co(NH3)6]Cl3, in which he distinguished principal and subsidiary valences (German: ‘Hauptvalenz’ and ‘Nebenvalenz’), corresponding to the modern concepts of oxidation state and coordination number respectively.
For main-group elements, in 1904 Richard Abegg considered positive and negative valences (maximal and minimal oxidation states), and proposed Abegg’s rule to the effect that their difference is often 8.
Electrons and valence
The Rutherford model of the nuclear atom (1911) showed that the exterior of an atom is occupied by electrons, which suggests that electrons are responsible for the interaction of atoms and the formation of chemical bonds. In 1916, Gilbert N. Lewis explained valence and chemical bonding in terms of a tendency of (main-group) atoms to achieve a stable octet of 8 valence-shell electrons. According to Lewis, covalent bonding leads to octets by the sharing of electrons, and ionic bonding leads to octets by the transfer of electrons from one atom to the other. The term covalence is attributed to Irving Langmuir, who stated in 1919 that «the number of pairs of electrons which any given atom shares with the adjacent atoms is called the covalence of that atom».[8] The prefix co- means «together», so that a co-valent bond means that the atoms share a valence. Subsequent to that, it is now more common to speak of covalent bonds rather than valence, which has fallen out of use in higher-level work from the advances in the theory of chemical bonding, but it is still widely used in elementary studies, where it provides a heuristic introduction to the subject.
In the 1930s, Linus Pauling proposed that there are also polar covalent bonds, which are intermediate between covalent and ionic, and that the degree of ionic character depends on the difference of electronegativity of the two bonded atoms.
Pauling also considered hypervalent molecules, in which main-group elements have apparent valences greater than the maximal of 4 allowed by the octet rule. For example, in the sulfur hexafluoride molecule (SF6), Pauling considered that the sulfur forms 6 true two-electron bonds using sp3d2 hybrid atomic orbitals, which combine one s, three p and two d orbitals. However more recently, quantum-mechanical calculations on this and similar molecules have shown that the role of d orbitals in the bonding is minimal, and that the SF6 molecule should be described as having 6 polar covalent (partly ionic) bonds made from only four orbitals on sulfur (one s and three p) in accordance with the octet rule, together with six orbitals on the fluorines.[9] Similar calculations on transition-metal molecules show that the role of p orbitals is minor, so that one s and five d orbitals on the metal are sufficient to describe the bonding.[10]
Common valences
For elements in the main groups of the periodic table, the valence can vary between 1 and 7.
| Group | Valence 1 | Valence 2 | Valence 3 | Valence 4 | Valence 5 | Valence 6 | Valence 7 | Valence 8 | Typical valences |
|---|---|---|---|---|---|---|---|---|---|
| 1 (I) | NaCl | 1 | |||||||
| 2 (II) | MgCl2 | 2 | |||||||
| 13 (III) | BCl3 AlCl3 Al2O3 | 3 | |||||||
| 14 (IV) | CO | CH4 | 4 | ||||||
| 15 (V) | NO | NH3 PH3 As2O3 | NO2 | N2O5 PCl5 | 3 and 5 | ||||
| 16 (VI) | H2O H2S | SO2 | SO3 | 2 and 6 | |||||
| 17 (VII) | HCl | HClO2 | ClO2 | HClO3 | Cl2O7 | 1 and 7 | |||
| 18 (VIII) | XeO4 | 8 |
Many elements have a common valence related to their position in the periodic table, and nowadays this is rationalised by the octet rule.
The Greek/Latin numeral prefixes (mono-/uni-, di-/bi-, tri-/ter-, and so on) are used to describe ions in the charge states 1, 2, 3, and so on, respectively. Polyvalence or multivalence refers to species that are not restricted to a specific number of valence bonds. Species with a single charge are univalent (monovalent). For example, the Cs+ cation is a univalent or monovalent cation, whereas the Ca2+ cation is a divalent cation, and the Fe3+ cation is a trivalent cation. Unlike Cs and Ca, Fe can also exist in other charge states, notably 2+ and 4+, and is thus known as a multivalent (polyvalent) ion.[11] Transition metals and metals to the right are typically multivalent but there is no simple pattern predicting their valency.[12]
| Valence | More common adjective‡ | Less common synonymous adjective‡§ |
|---|---|---|
| 0-valent | zerovalent | nonvalent |
| 1-valent | monovalent | univalent |
| 2-valent | divalent | bivalent |
| 3-valent | trivalent | tervalent |
| 4-valent | tetravalent | quadrivalent |
| 5-valent | pentavalent | quinquevalent, quinquivalent |
| 6-valent | hexavalent | sexivalent |
| 7-valent | heptavalent | septivalent |
| 8-valent | octavalent | — |
| 9-valent | nonavalent | — |
| 10-valent | decavalent | — |
| 12-valent | dodecavalent | — |
| multiple / many / variable | polyvalent | multivalent |
| together | covalent | — |
| not together | noncovalent | — |
† The same adjectives are also used in medicine to refer to vaccine valence, with the slight difference that in the latter sense, quadri- is more common than tetra-.
‡ As demonstrated by hit counts in Google web search and Google Books search corpora (accessed 2017).
§ A few other forms can be found in large English-language corpora (for example, *quintavalent, *quintivalent, *decivalent), but they are not the conventionally established forms in English and thus are not entered in major dictionaries.
Valence versus oxidation state
Because of the ambiguity of the term valence,[13] other notations are currently preferred. Beside the system of oxidation states (also called oxidation numbers) as used in Stock nomenclature for coordination compounds,[14] and the lambda notation, as used in the IUPAC nomenclature of inorganic chemistry,[15] oxidation state is a more clear indication of the electronic state of atoms in a molecule.
The oxidation state of an atom in a molecule gives the number of valence electrons it has gained or lost.[16] In contrast to the valency number, the oxidation state can be positive (for an electropositive atom) or negative (for an electronegative atom).
Elements in a high oxidation state have an oxidation state higher than +4, and also, elements in a high valence state (hypervalent elements) have a valence higher than 4. For example, in perchlorates ClO−4, chlorine has 7 valence bonds (thus, it is heptavalent, in other words, it has valence 7), and it has oxidation state +7; in ruthenium tetroxide RuO4, ruthenium has 8 valence bonds (thus, it is octavalent, in other words, it has valence 8), and it has oxidation state +8.
In some scenarios, the difference between valence and oxidation state arises. Valence and oxidation state of the same atom may not be the same. For example, in disulfur decafluoride molecule S2F10, each sulfur atom has 6 valence bonds (5 single bonds with fluorine atoms and 1 single bond with sulfur atom), thus, each sulfur atom is hexavalent, in other words, it has valence 6, but has oxidation state +5. In dioxygen molecule O2, each oxygen atom has 2 valence bonds, thus, each oxygen atom is divalent, in other words, it has valence 2, but has oxidation state 0. In acetylene H−C≡C−H, each carbon atom has 4 valence bonds (1 single bond with hydrogen atom and 3 single bonds with carbon atom), thus, each carbon atom is tetravalent, in other words, it has valence 4, but has oxidation state −1.
Examples
| Compound | Formula | Valence | Oxidation state | Diagram |
|---|---|---|---|---|
| Hydrogen chloride | HCl | H = 1 Cl = 1 | H = +1 Cl = −1 | H−Cl |
| Perchloric acid * | HClO4 | H = 1 Cl = 7 O = 2 | H = +1 Cl = +7 O = −2 |  |
| Methane | CH4 | C = 4 H = 1 | C = −4 H = +1 |  |
| Dichloromethane ** | CH2Cl2 | C = 4 H = 1 Cl = 1 | C = 0 H = +1 Cl = −1 |  |
| Ferrous oxide *** | FeO | Fe = 2 O = 2 | Fe = +2 O = −2 | Fe=O |
| Ferric oxide *** | Fe2O3 | Fe = 3 O = 2 | Fe = +3 O = −2 | O=Fe−O−Fe=O |
| Sodium hydride | NaH | Na = 1 H = 1 | Na = +1 H = −1 | Na−H |
* The perchlorate ion ClO−4 is monovalent, in other words, it has valence 1.
** Valences may also be different from absolute values of oxidation states due to different polarity of bonds. For example, in dichloromethane, CH2Cl2, carbon has valence 4 but oxidation state 0.
*** Iron oxides appear in a crystal structure, so no typical molecule can be identified. In ferrous oxide, Fe has oxidation state +2; in ferric oxide, oxidation state +3.
| Compound | Formula | Valence | Oxidation state | Diagram |
|---|---|---|---|---|
| Hydrogen | H2 | H = 1 | H = 0 | H−H |
| Chlorine | Cl2 | Cl = 1 | Cl = 0 | Cl−Cl |
| Hydrogen peroxide | H2O2 | H = 1 O = 2 | H = +1 O = −1 |  |
| Hydrazine | N2H4 | H = 1 N = 3 | H = +1 N = −2 |  |
| Disulfur decafluoride | S2F10 | S = 6 F = 1 | S = +5 F = −1 |  |
| Dithionic acid | H2S2O6 | S = 6 O = 2 H = 1 | S = +5 O = −2 H = +1 |  |
| Hexachloroethane | C2Cl6 | C = 4 Cl = 1 | C = +3 Cl = −1 |  |
| Ethylene | C2H4 | C = 4 H = 1 | C = −2 H = +1 |  |
| Acetylene | C2H2 | C = 4 H = 1 | C = −1 H = +1 | H−C≡C−H |
| Mercury(I) chloride | Hg2Cl2 | Hg = 2 Cl = 1 | Hg = +1 Cl = −1 | Cl−Hg−Hg−Cl |
«Maximum number of bonds» definition
Frankland took the view that the valence (he used the term «atomicity») of an element was a single value that corresponded to the maximum value observed. The number of unused valencies on atoms of what are now called the p-block elements is generally even, and Frankland suggested that the unused valencies saturated one another. For example, nitrogen has a maximum valence of 5, in forming ammonia two valencies are left unattached; sulfur has a maximum valence of 6, in forming hydrogen sulphide four valencies are left unattached.[17][18]
The International Union of Pure and Applied Chemistry (IUPAC) has made several attempts to arrive at an unambiguous definition of valence. The current version, adopted in 1994:[19]
- The maximum number of univalent atoms (originally hydrogen or chlorine atoms) that may combine with an atom of the element under consideration, or with a fragment, or for which an atom of this element can be substituted.[2]
Hydrogen and chlorine were originally used as examples of univalent atoms, because of their nature to form only one single bond. Hydrogen has only one valence electron and can form only one bond with an atom that has an incomplete outer shell. Chlorine has seven valence electrons and can form only one bond with an atom that donates a valence electron to complete chlorine’s outer shell. However, chlorine can also have oxidation states from +1 to +7 and can form more than one bond by donating valence electrons.
Hydrogen has only one valence electron, but it can form bonds with more than one atom. In the bifluoride ion ([HF2]−), for example, it forms a three-center four-electron bond with two fluoride atoms:
- [F−H F− ↔ F− H−F]
Another example is the three-center two-electron bond in diborane (B2H6).
Maximum valences of the elements
Maximum valences for the elements are based on the data from list of oxidation states of the elements. They are shown by the color code at the bottom of the table.
Maximum valences of the elements | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | ||
| Group → | |||||||||||||||||||
| ↓ Period | |||||||||||||||||||
| 1 | 1 H | 2 He | |||||||||||||||||
| 2 | 3 Li | 4 Be | 5 B | 6 C | 7 N | 8 O | 9 F | 10 Ne | |||||||||||
| 3 | 11 Na | 12 Mg | 13 Al | 14 Si | 15 P | 16 S | 17 Cl | 18 Ar | |||||||||||
| 4 | 19 K | 20 Ca | 21 Sc | 22 Ti | 23 V | 24 Cr | 25 Mn | 26 Fe | 27 Co | 28 Ni | 29 Cu | 30 Zn | 31 Ga | 32 Ge | 33 As | 34 Se | 35 Br | 36 Kr | |
| 5 | 37 Rb | 38 Sr | 39 Y | 40 Zr | 41 Nb | 42 Mo | 43 Tc | 44 Ru | 45 Rh | 46 Pd | 47 Ag | 48 Cd | 49 In | 50 Sn | 51 Sb | 52 Te | 53 I | 54 Xe | |
| 6 | 55 Cs | 56 Ba | 71 Lu | 72 Hf | 73 Ta | 74 W | 75 Re | 76 Os | 77 Ir | 78 Pt | 79 Au | 80 Hg | 81 Tl | 82 Pb | 83 Bi | 84 Po | 85 At | 86 Rn | |
| 7 | 87 Fr | 88 Ra | 103 Lr | 104 Rf | 105 Db | 106 Sg | 107 Bh | 108 Hs | 109 Mt | 110 Ds | 111 Rg | 112 Cn | 113 Nh | 114 Fl | 115 Mc | 116 Lv | 117 Ts | 118 Og | |
| 57 La | 58 Ce | 59 Pr | 60 Nd | 61 Pm | 62 Sm | 63 Eu | 64 Gd | 65 Tb | 66 Dy | 67 Ho | 68 Er | 69 Tm | 70 Yb | ||||||
| 89 Ac | 90 Th | 91 Pa | 92 U | 93 Np | 94 Pu | 95 Am | 96 Cm | 97 Bk | 98 Cf | 99 Es | 100 Fm | 101 Md | 102 No | ||||||
| Maximum valences are based on the List of oxidation states of the elements | |||||||||||||||||||
|
Primordial From decay Synthetic Border shows natural occurrence of the element |
See also
- Abegg’s rule
- Oxidation state
References
- ^ a b Partington, James Riddick (1921). A text-book of inorganic chemistry for university students (1st ed.). OL 7221486M.
- ^ a b IUPAC Gold Book definition: valence
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ^ Parkin, Gerard (May 2006). «Valence, Oxidation Number, and Formal Charge: Three Related but Fundamentally Different Concepts». Journal of Chemical Education. 83 (5): 791. doi:10.1021/ed083p791. ISSN 0021-9584.
- ^ Harper, Douglas. «valence». Online Etymology Dictionary.
- ^ a b Partington, J.R. (1989). A Short History of Chemistry. Dover Publications, Inc. ISBN 0-486-65977-1.
- ^ Frankland, E. (1852). «On a New Series of Organic Bodies Containing Metals». Philosophical Transactions of the Royal Society of London. 142: 417–444. doi:10.1098/rstl.1852.0020. S2CID 186210604.
- ^ Langmuir, Irving (1919). «The Arrangement of Electrons in Atoms and Molecules». Journal of the American Chemical Society. 41 (6): 868–934. doi:10.1021/ja02227a002.
- ^ Magnusson, Eric (1990). «Hypercoordinate molecules of second-row elements: d functions or d orbitals?». J. Am. Chem. Soc. 112 (22): 7940–7951. doi:10.1021/ja00178a014.
- ^ Frenking, Gernot; Shaik, Sason, eds. (May 2014). «Chapter 7: Chemical bonding in Transition Metal Compounds». The Chemical Bond: Chemical Bonding Across the Periodic Table. Wiley – VCH. ISBN 978-3-527-33315-8.
- ^ Merriam-Webster, Merriam-Webster’s Unabridged Dictionary, Merriam-Webster, archived from the original on 2020-05-25, retrieved 2017-05-11.
- ^ «Lesson 7: Ions and Their Names». Clackamas Community College. Archived from the original on 21 January 2019. Retrieved 5 February 2019.
- ^ The Free Dictionary: valence
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the «Gold Book») (1997). Online corrected version: (2006–) «Oxidation number». doi:10.1351/goldbook.O04363
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the «Gold Book») (1997). Online corrected version: (2006–) «Lambda». doi:10.1351/goldbook.L03418
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the «Gold Book») (1997). Online corrected version: (2006–) «Oxidation state». doi:10.1351/goldbook.O04365
- ^ Frankland, E. (1870). Lecture notes for chemical students(Google eBook) (2d ed.). J. Van Voorst. p. 21.
- ^ Frankland, E.; Japp, F.R (1885). Inorganic chemistry (1st ed.). pp. 75–85. OL 6994182M.
- ^ Muller, P. (1994). «Glossary of terms used in physical organic chemistry (IUPAC Recommendations 1994)». Pure and Applied Chemistry. 66 (5): 1077–1184. doi:10.1351/pac199466051077. S2CID 195819485.

